Answer
386.7k+ views
Hint: We can say that another name of canonical structure is resonance structure, i.e. one of possibly more than one contributing structures which combine to form the true, resonance hybrid structure. We can say resonance is a way of explaining bonding in some molecules (or) ions by combining many contributing structures into a resonance hybrid.
Complete step by step answer:
We have to know that canonical structure is a way of indicating (or) explaining the delocalized electrons inside several molecules (or) polyatomic ions such as sulfate, phosphate ions where the bonding could not be represented based on Lewis structures. Resonance is the delocalization of pi electrons that are presented in several intermediate structures known as canonical forms.
The resonance hybrid represents the actual molecule as the average of contributing structures with partial charges and bond length taking on intermediate values compared to those for the individual Lewis structure of the contributors where they are found as real chemical entities.
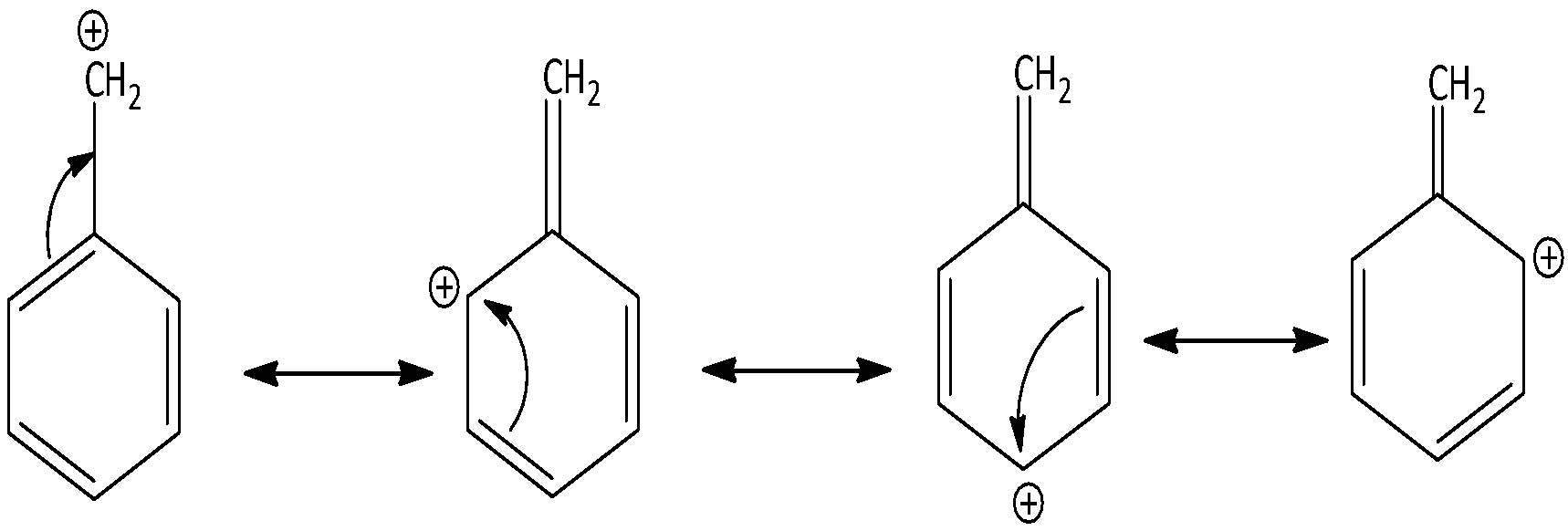
Contributing structures are separated by double headed arrows. In a hybrid structure, pi bonds are involved in resonance.
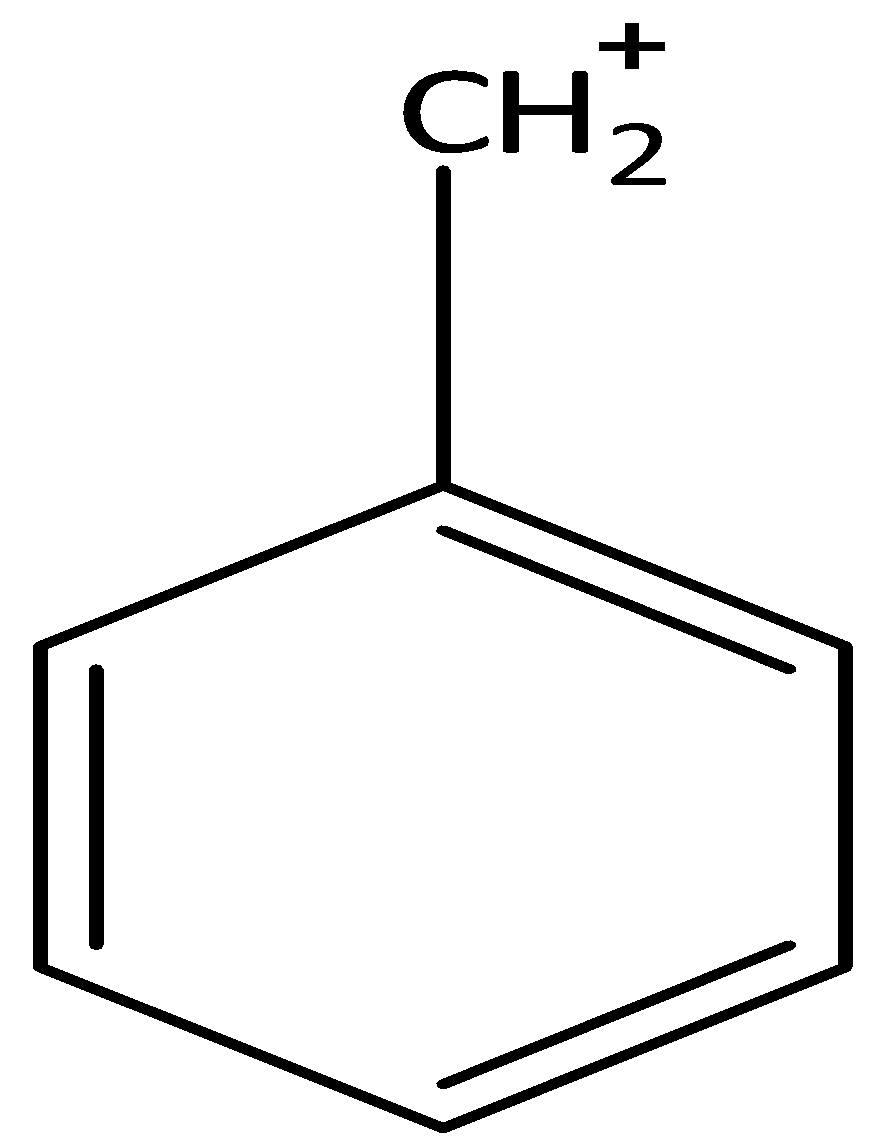
There are four resonance forms of benzyl carbocation ${C_6}{H_5} - C{H_2}^ + $. We can draw the canonical forms of benzyl carbocation as,
Molecules that have extended pi systems like linear polyenes and polyaromatic compounds are explained by both resonance hydrides and delocalized orbitals in molecular orbital theory.
Note: In other aromatic rings and benzene, a solid circle is used to indicate the delocalized pi electrons. We have to know that resonances and isomerism are two different concepts. We know that isomers are molecules that have the same chemical formula but different in spatial arrangements of atoms. Resonance differs in the way electrons are formally assigned to atoms in Lewis structure descriptions of the molecule.
Complete step by step answer:
We have to know that canonical structure is a way of indicating (or) explaining the delocalized electrons inside several molecules (or) polyatomic ions such as sulfate, phosphate ions where the bonding could not be represented based on Lewis structures. Resonance is the delocalization of pi electrons that are presented in several intermediate structures known as canonical forms.
The resonance hybrid represents the actual molecule as the average of contributing structures with partial charges and bond length taking on intermediate values compared to those for the individual Lewis structure of the contributors where they are found as real chemical entities.
Contributing structures are separated by double headed arrows. In a hybrid structure, pi bonds are involved in resonance.
There are four resonance forms of benzyl carbocation ${C_6}{H_5} - C{H_2}^ + $. We can draw the canonical forms of benzyl carbocation as,

Molecules that have extended pi systems like linear polyenes and polyaromatic compounds are explained by both resonance hydrides and delocalized orbitals in molecular orbital theory.
Note: In other aromatic rings and benzene, a solid circle is used to indicate the delocalized pi electrons. We have to know that resonances and isomerism are two different concepts. We know that isomers are molecules that have the same chemical formula but different in spatial arrangements of atoms. Resonance differs in the way electrons are formally assigned to atoms in Lewis structure descriptions of the molecule.
Recently Updated Pages
Basicity of sulphurous acid and sulphuric acid are

Three beakers labelled as A B and C each containing 25 mL of water were taken A small amount of NaOH anhydrous CuSO4 and NaCl were added to the beakers A B and C respectively It was observed that there was an increase in the temperature of the solutions contained in beakers A and B whereas in case of beaker C the temperature of the solution falls Which one of the following statements isarecorrect i In beakers A and B exothermic process has occurred ii In beakers A and B endothermic process has occurred iii In beaker C exothermic process has occurred iv In beaker C endothermic process has occurred

What is the stopping potential when the metal with class 12 physics JEE_Main

The momentum of a photon is 2 times 10 16gm cmsec Its class 12 physics JEE_Main

How do you arrange NH4 + BF3 H2O C2H2 in increasing class 11 chemistry CBSE

Is H mCT and q mCT the same thing If so which is more class 11 chemistry CBSE

Trending doubts
Difference Between Plant Cell and Animal Cell

Difference between Prokaryotic cell and Eukaryotic class 11 biology CBSE

Fill the blanks with the suitable prepositions 1 The class 9 english CBSE

Change the following sentences into negative and interrogative class 10 english CBSE

Give 10 examples for herbs , shrubs , climbers , creepers

Fill the blanks with proper collective nouns 1 A of class 10 english CBSE

Select the word that is correctly spelled a Twelveth class 10 english CBSE

How fast is 60 miles per hour in kilometres per ho class 10 maths CBSE

What organs are located on the left side of your body class 11 biology CBSE